
Picking HVAC Guidance Needles in PIC/S Haystack
FDA GMP regulations are legally binding for drugs marketed in the US. Similarly, EU GMP regulations are binding for the EU market. But what about the rest of the world?
That’s where PIC/S (Pharmaceutical Inspection Co-operation Scheme) comes in. While not legally binding, it offers harmonized guidance that over 40 countries follow.
PIC/S is an international cooperative arrangement among regulatory authorities in the field of Good Manufacturing Practice (GMP) for medicines and active pharmaceutical ingredients (APIs).
The main objective of PIC/S is to harmonize GMP standards and inspection procedures globally and to facilitate cooperation and mutual recognition among health authorities.
But for our purpose, the key question is:
What does PIC/S say about HVAC systems in the context of sterile manufacturing?
FIRST, THE HAYSTACK
Before we find the HVAC guidance needles in the PIC/S haystack (nearly 500 pages), let’s get familiar with the core PIC/S GMP documents. The latest version (dated 25 August 2023) includes:
- PE 009-17 (Intro): Guide to GMP for Medicinal Products
- PE 009-17 (Part I): GMP for Medicinal Products – Part I
- PE 009-17 (Part II): GMP for APIs
- PE 009-17 (Annexes): GMP Annexes
NOW, THE HVAC NEEDLES
After scanning all four documents, here are the key HVAC-related requirements and best practices that one should keep in mind while designing HVAC systems:
1. HVAC zoning
- Keep Air Handling Unit (AHU) zoning separate for production areas. Avoid mixing with non-production zones.
2. Cleanroom classification
- Perform classification in both “at rest” and “in operation” states.
- Follow defined maximum permitted total particle concentrations for each grade. (See the table below.)
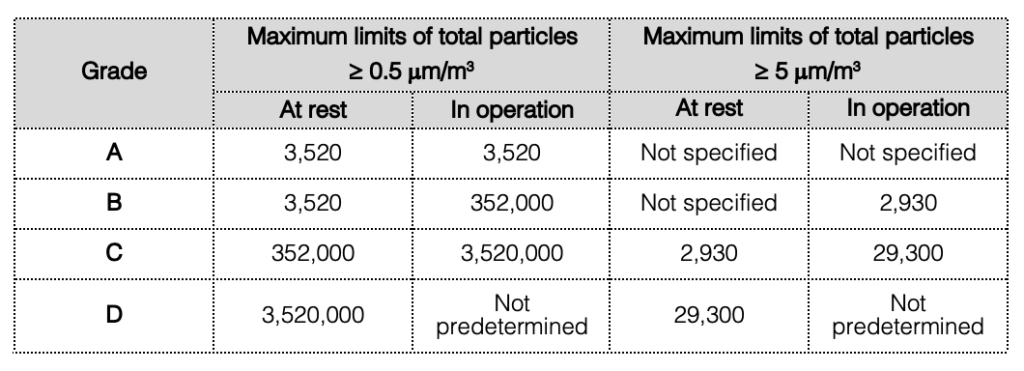
3. Pressure differential
- Maintain positive pressure relative to lower-grade rooms or background areas.
- Target a minimum differential pressure of 10 Pascals between adjacent rooms of different grades.
- Integrate pressure setpoints and criticality within the Contamination Control Strategy (CCS).
4. Pressure monitoring, recording & alarming
- Install differential pressure indicators between cleanrooms (or isolators and background).
- Continuously monitor and record critical pressure differences.
- Set up alarm systems to instantly warn operators of air supply failures or critical pressure drops.
- Alarms should not be overridden without following an approved procedure and documented assessment.
- Any alarm delay should be assessed and justified in the CCS.
5. Decontamination
- Consider decontamination of cleanrooms and HVAC systems, especially when dealing with high-risk operations.
- Consider air treatment measures for air leaving clean areas where applicable.
6. Airflow patterns
- Conduct airflow visualization studies to:
- Ensure no ingress from lower- to higher-grade areas (across cleanrooms and small egress points to transfer materials)
- Avoid air movement from less clean zones (e.g., floor, operator, equipment) to over cleaner areas.
- Where unidirectional airflow is used:
- Perform visualization studies to confirm compliance.
- Simulate operator interventions and other dynamic conditions.
- Document the results and retain video recordings.
- Use the findings to shape the environmental monitoring program.
7. Unidirectional Flow (UDF)
- Air velocity at working positions should be 0.36 – 0.54 m/s (guidance range), unless scientifically justified in the CCS.
- Justify air velocity and measurement locations in the qualification protocol.
8. Grade A Monitoring
- Monitor Grade A areas continuously for ≥0.5 µm and ≥5.0 µm particles.
- Use a suitable sample flow rate (minimum 28 L/min or 1 ft³/min) to capture:
- Interventions
- Transient events
- System deterioration
PIC/S may not be legally binding, but when over 40 countries align with its guidance, it becomes the closest thing to a global GMP benchmark. For HVAC designers and validation engineers already familiar with EU GMP, PIC/S holds no surprises—and that’s a good thing. It’s reassuring to know there are no special HVAC-specific clauses buried in the PIC/S haystack of guidance documents.
PIC/S GMP link: PIC/S GMP Guide (PE009-17) – Introduction, Part I, Part II & Annexes
Upcoming…
I will be offering a training program titled “HVAC Design & Qualification for Biopharma Facilities” (20 & 21 Aug. 2025). Check it out here.
